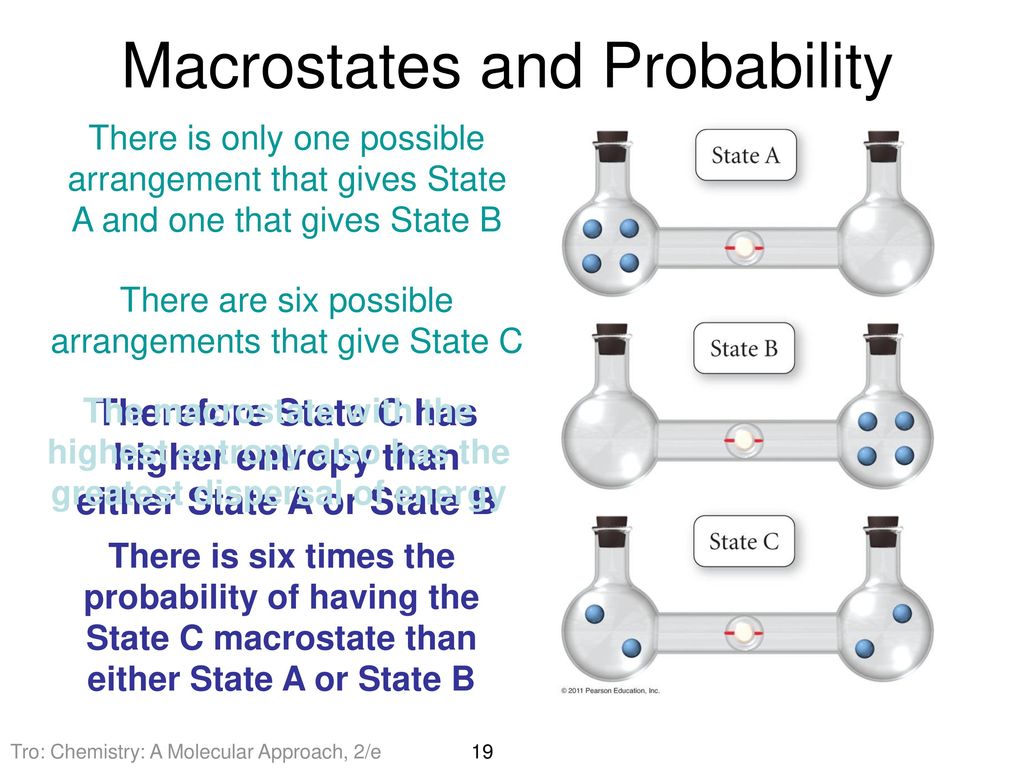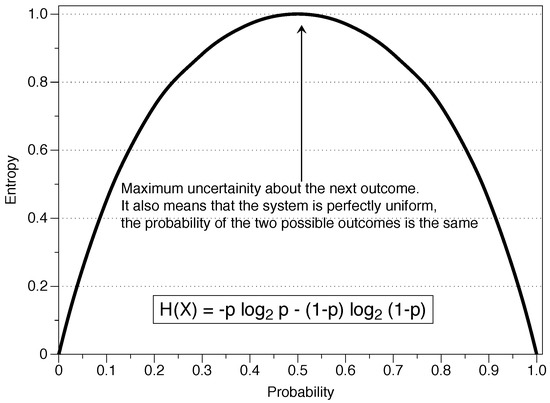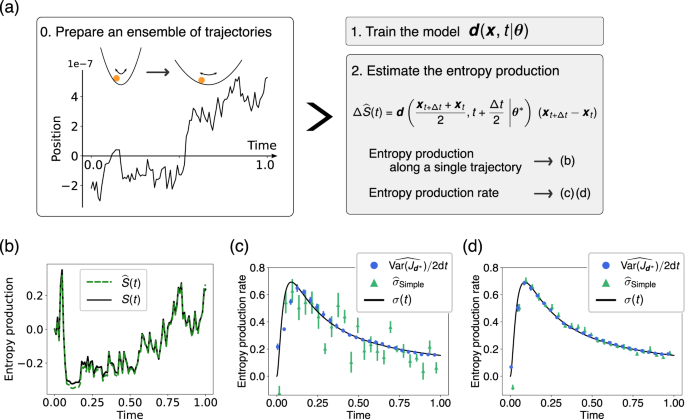
Estimating time-dependent entropy production from non-equilibrium trajectories | Communications Physics

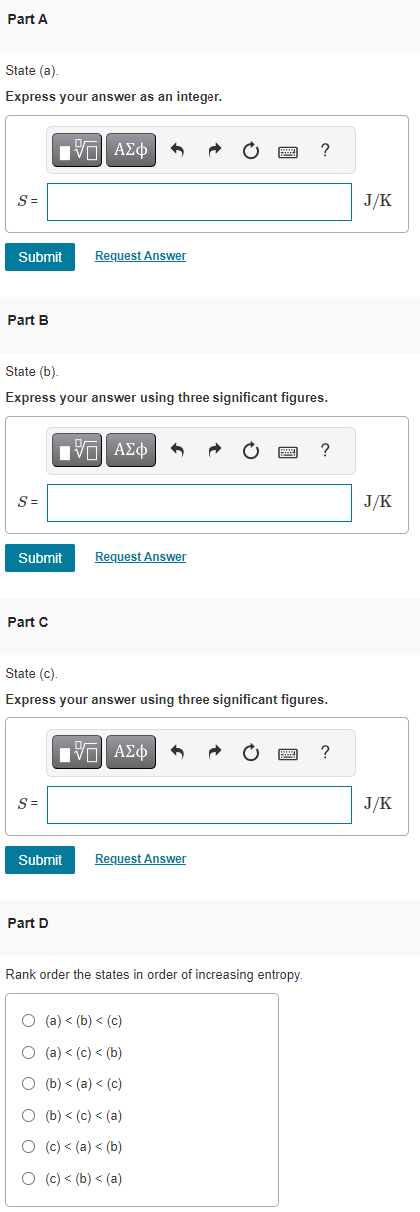
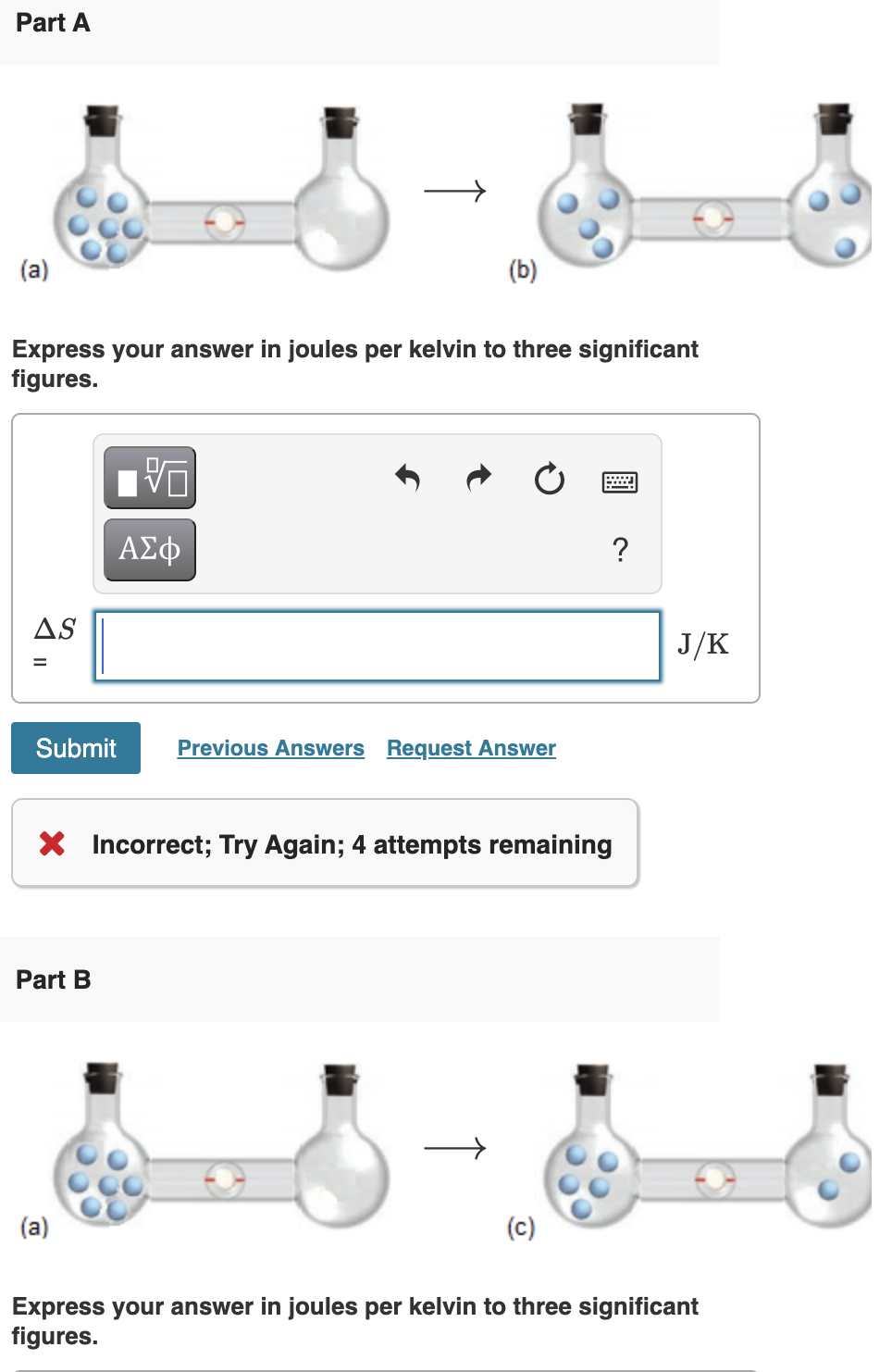
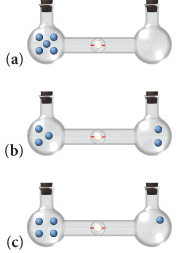
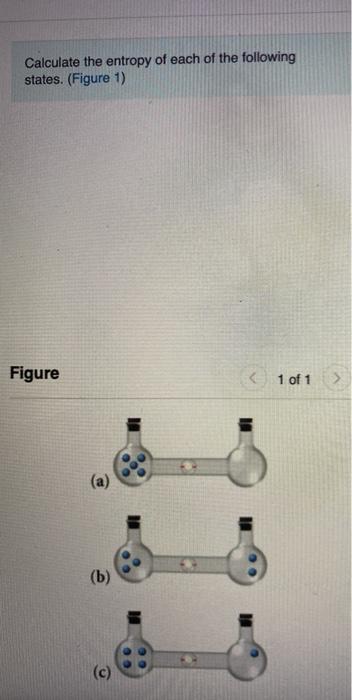
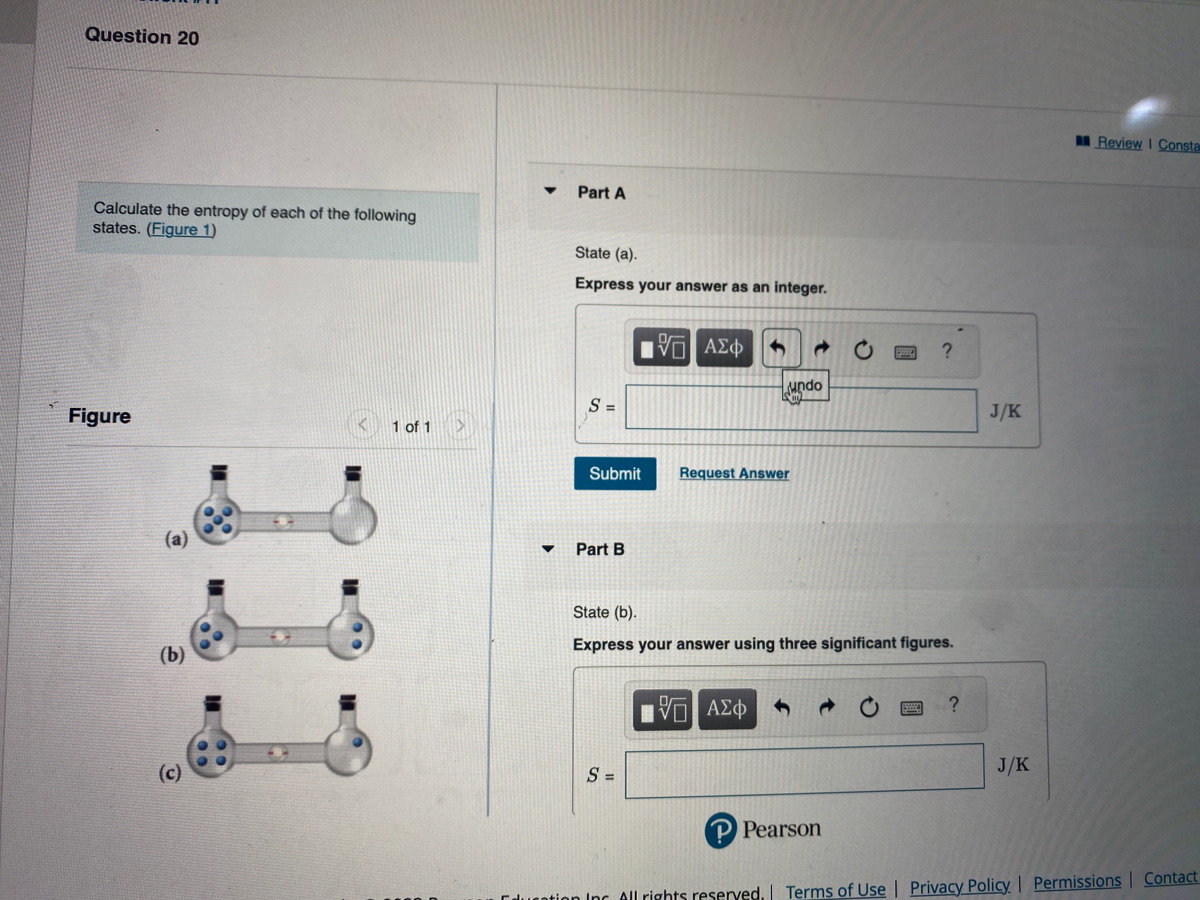
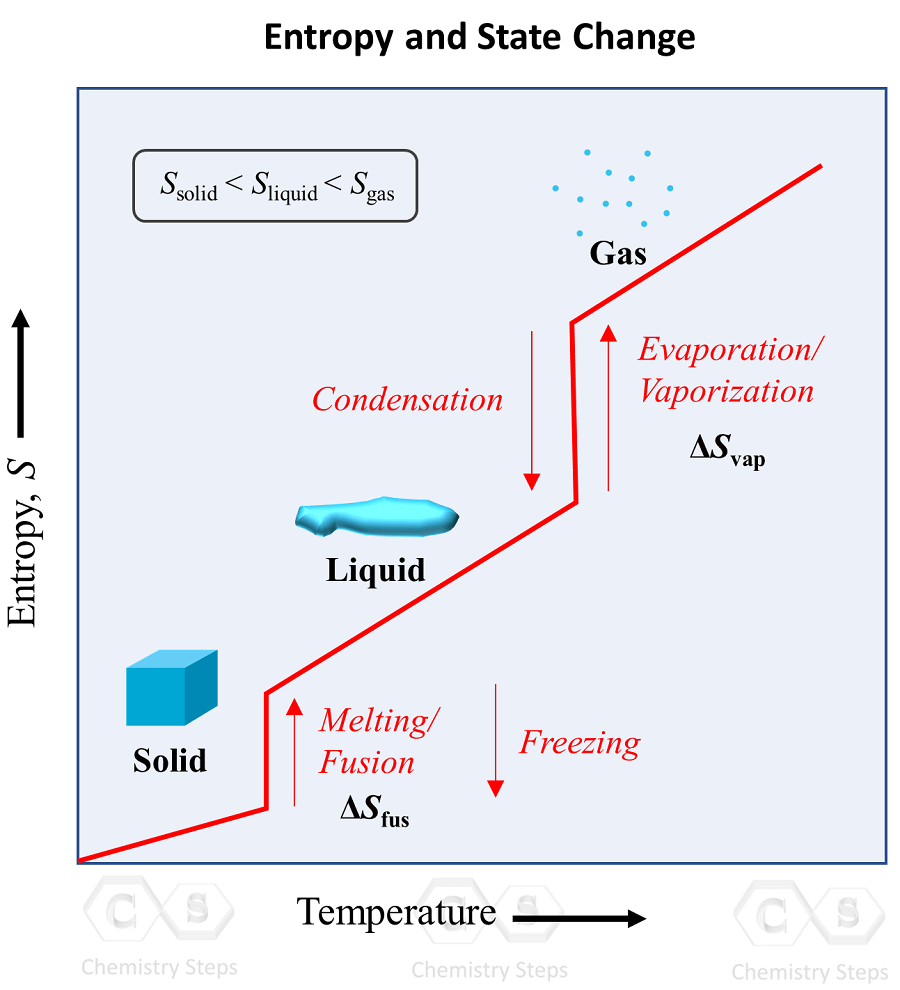
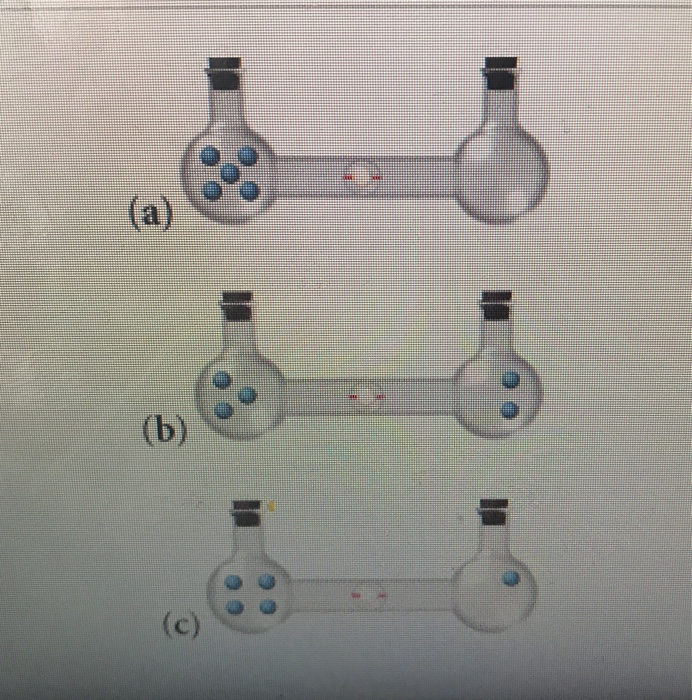
SOLVED:Calculate the entropy of each state and rank the states in order of increasing entropy. a. b. c.

SOLVED: Calculate the standard-state entropy for the following reaction: CHa(g) + 2 O2(g) = COz(g) + 2 Hz0(1) (If applicable, coefficients of one have been included for clarity:) The standard entropy values

SOLVED:Find the entropy for the following water states and indicate each state on a T-s diagram relative to the two-phase region. a. 250^∘ C, v=0.02 m^3 / kg b. 250^∘ C, 2000
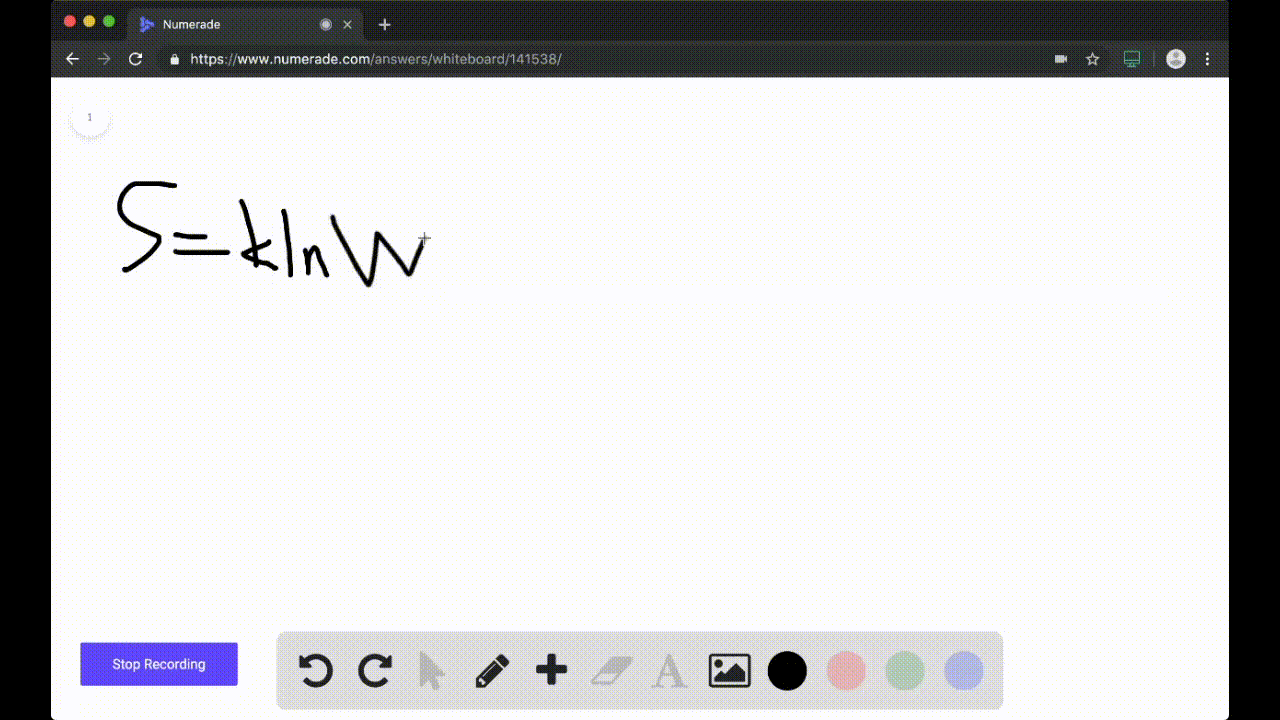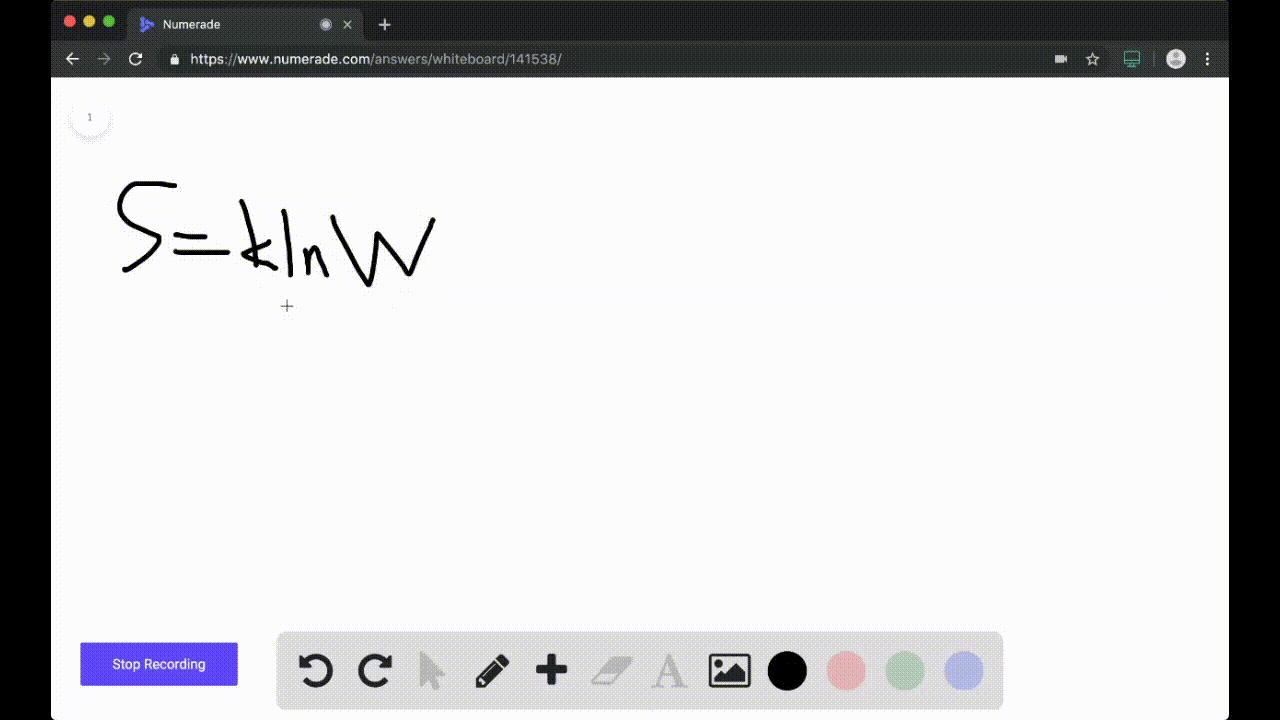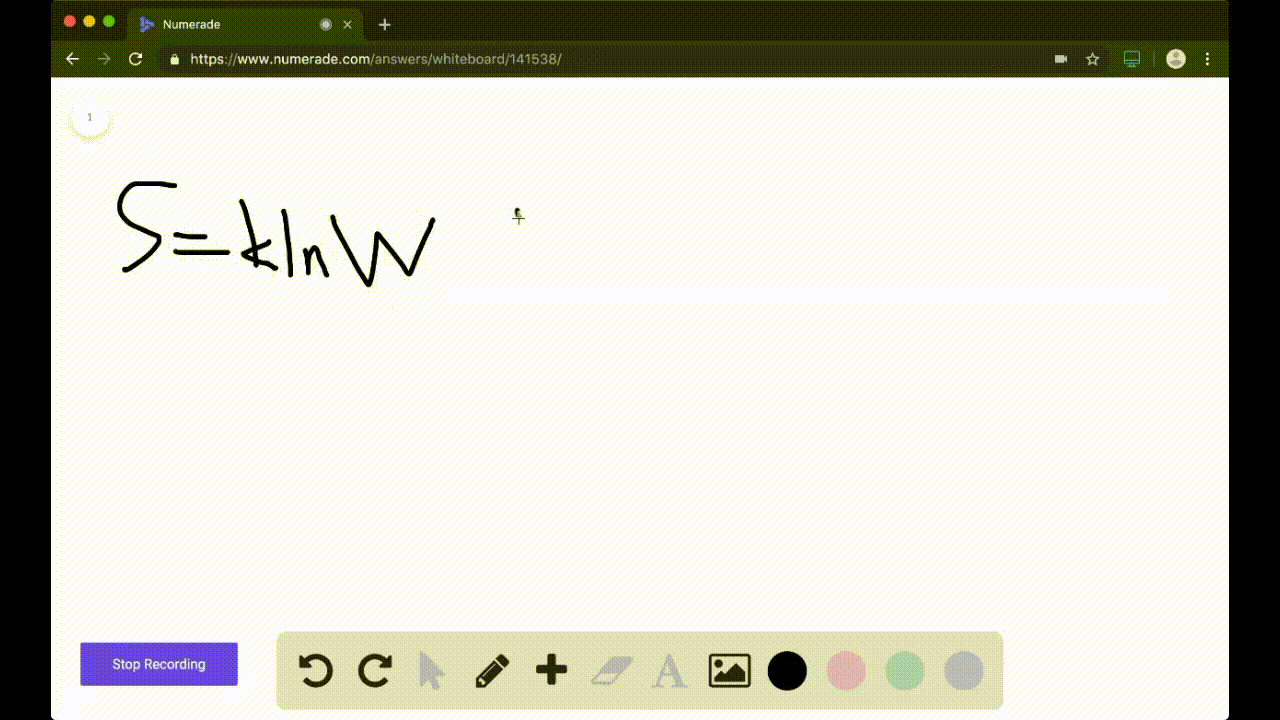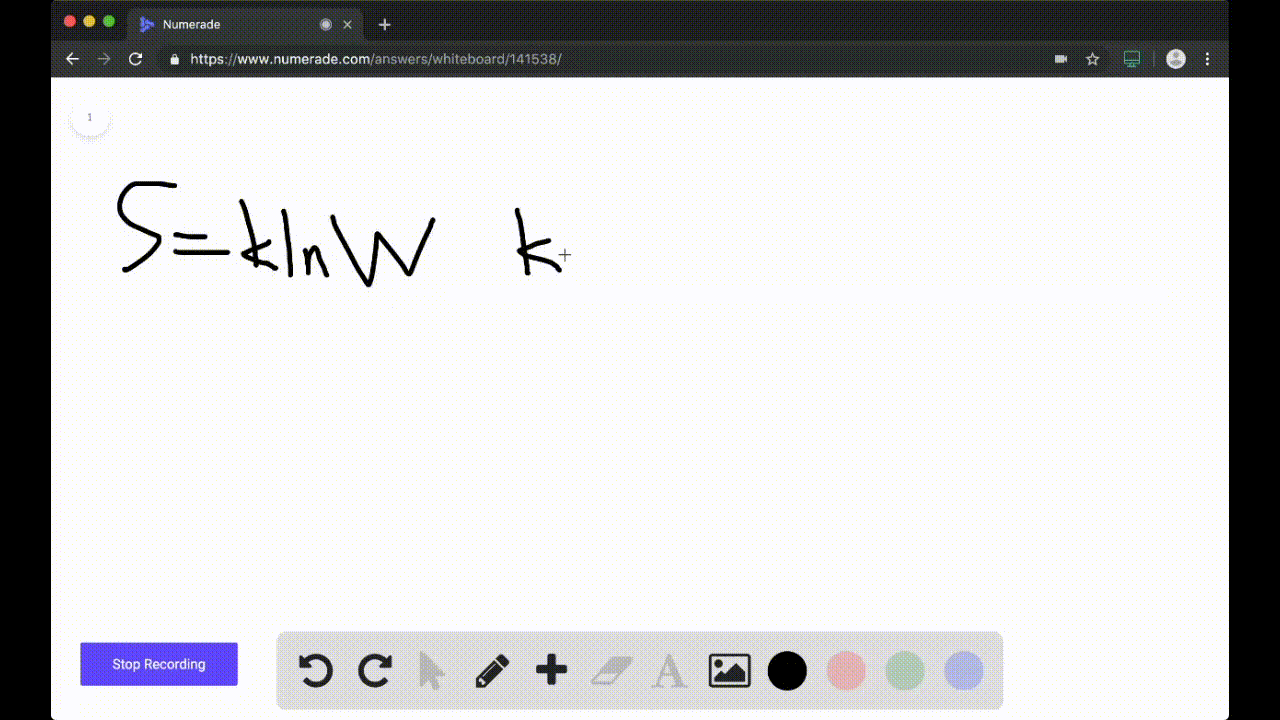
SOLVED:Calculate the entropy of each state and rank the states in order of increasing entropy. a. b. c.

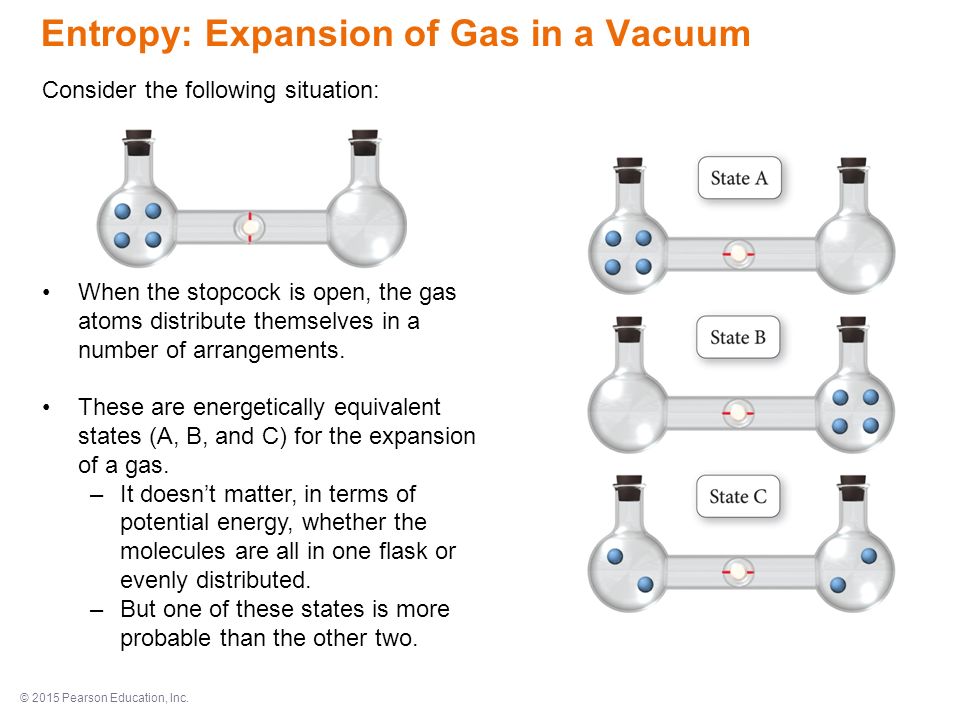
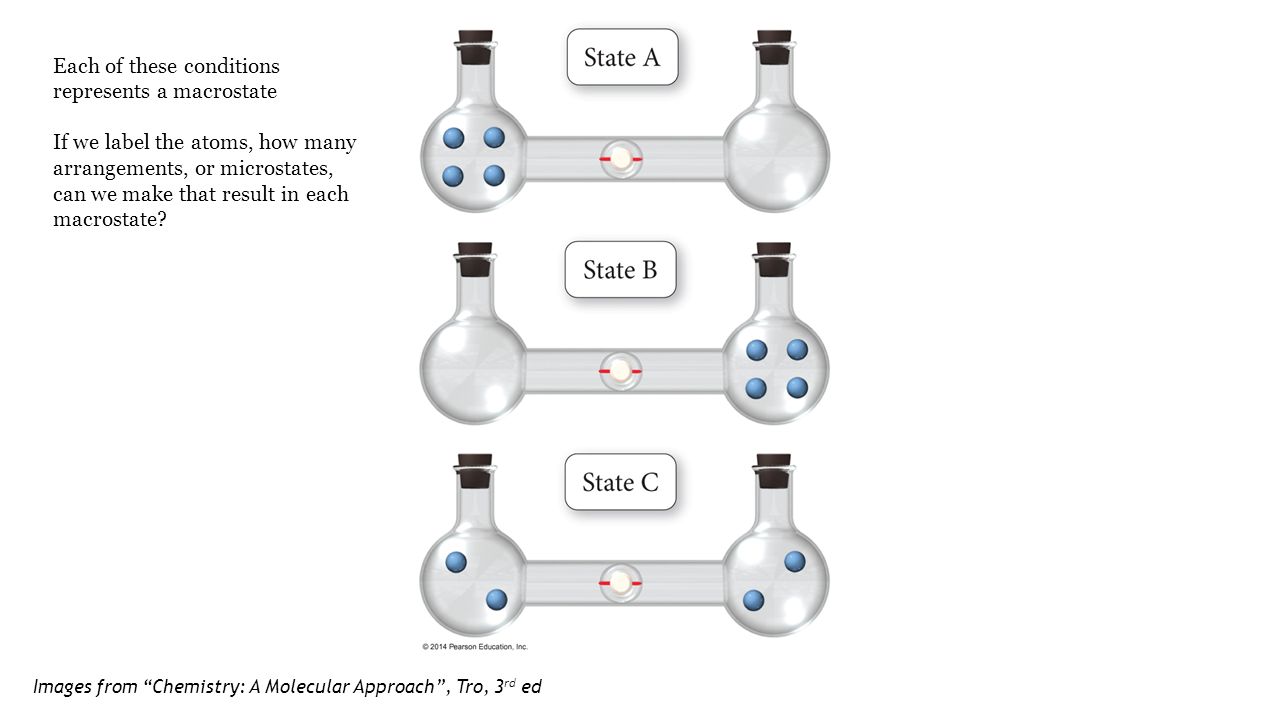
SOLVED:Consider the distribution of ideal gas molecules among three bulbs (A, B, and C) of equal volume. For each of the following states, determine the number of ways that the state can